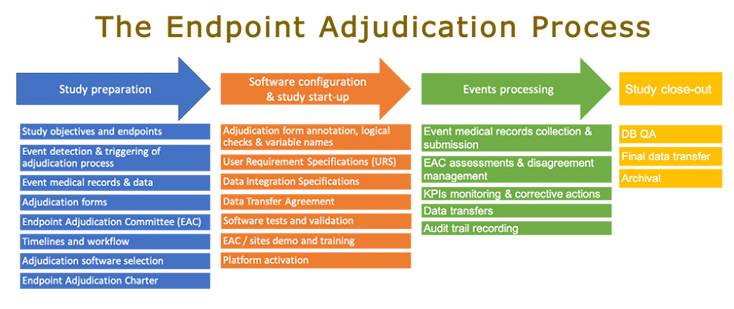
Reproducibility of clinical events adjudications in a trial of venous thromboembolism prevention - Girard - 2017 - Journal of Thrombosis and Haemostasis - Wiley Online Library

Debriefing in the Emergency Department After Clinical Events: A Practical Guide - Annals of Emergency Medicine

Blood-Based Cardiac Biomarkers and the Risk of Cognitive Decline, Cerebrovascular Disease, and Clinical Events | Stroke
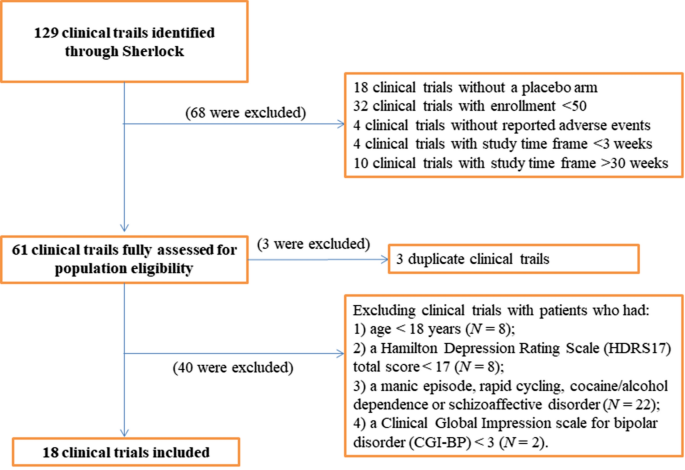
Identifying Anticipated Events of Future Clinical Trials by Leveraging Data from the Placebo Arms of Completed Trials | SpringerLink

Debriefing Real Clinical Events | Center for Advanced Pediatric & Perinatal Education (CAPE) | Stanford Medicine

Investigator-Reported Versus Adjudicated Clinical Events: 2 Versions of the Truth?∗ | Journal of the American College of Cardiology

Predicting severe clinical events by learning about life-saving actions and outcomes using distant supervision - ScienceDirect
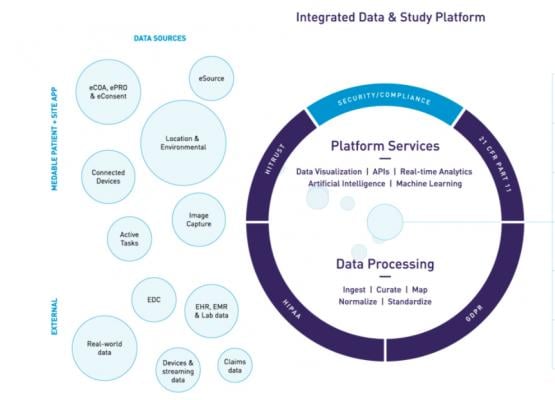
Virtual Study to Track Statin-Related Adverse Events Being Undertaken by University of Connecticut and Hartford Healthcare | DAIC

Major adverse clinical events associated with implantation of a leadless intracardiac pacemaker - Heart Rhythm

Clinical events classification (CEC) in clinical trials: Report on the current landscape and future directions — proceedings from the CEC Summit 2018 - ScienceDirect
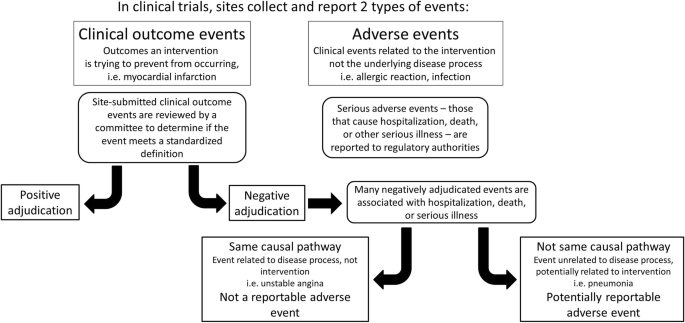
Methods for safety and endpoint ascertainment: identification of adverse events through scrutiny of negatively adjudicated events | Trials | Full Text