Amazon.co.jp: Gait Biometrics: Basic Patterns, Role of Neurological Disorders and Effects of Physical Activity (Physiology - Laboratory and Clinical Research) : Li, Li, Holmes, Matthew: Foreign Language Books

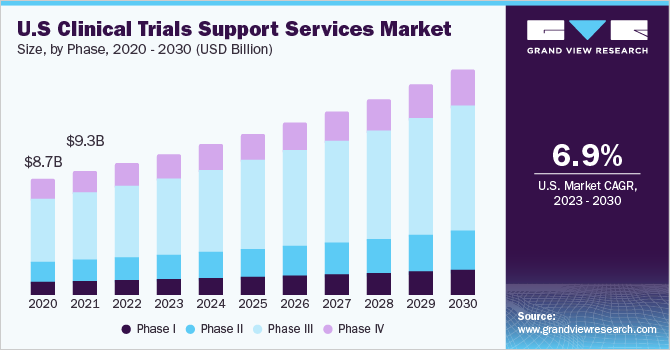
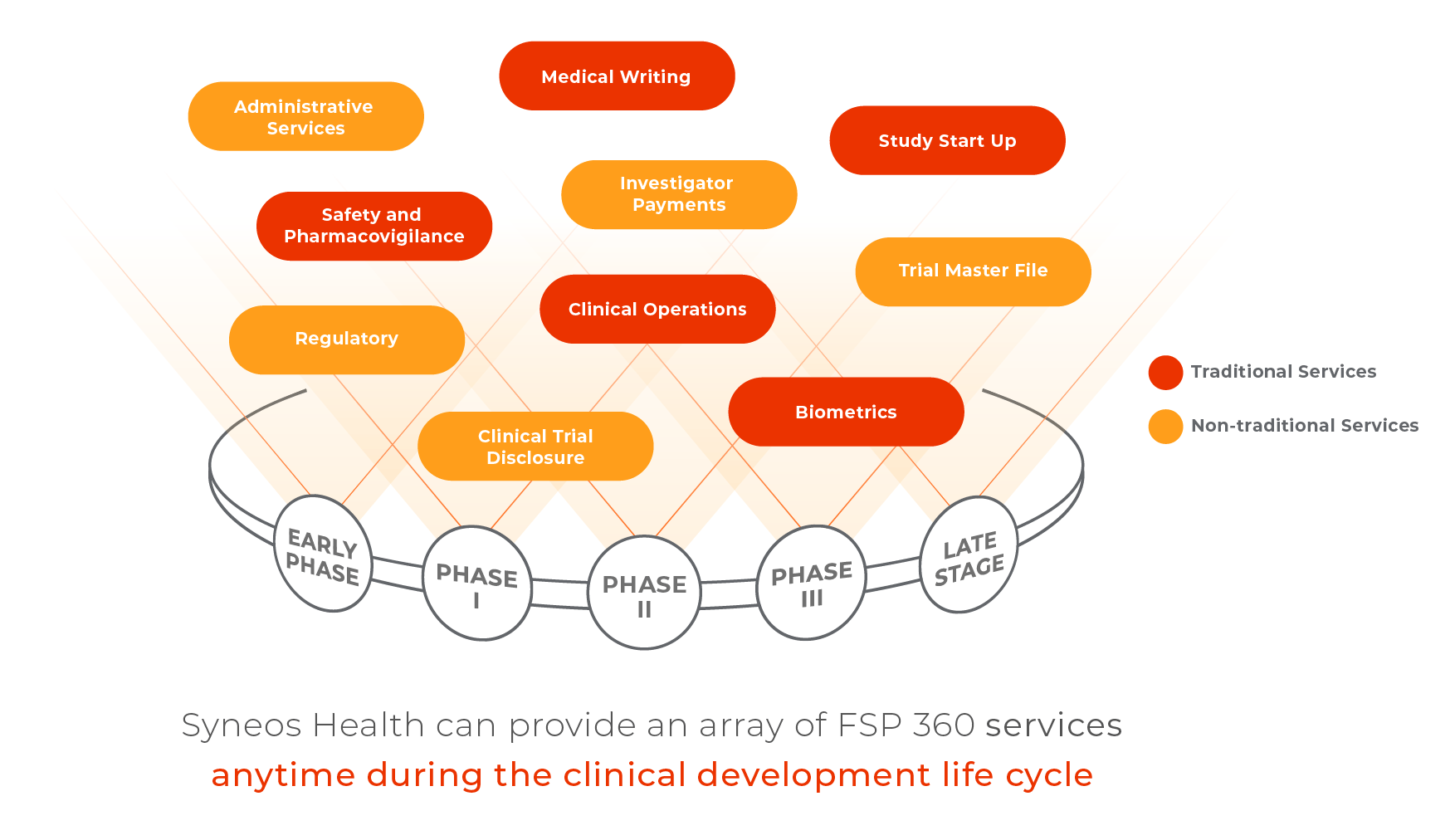
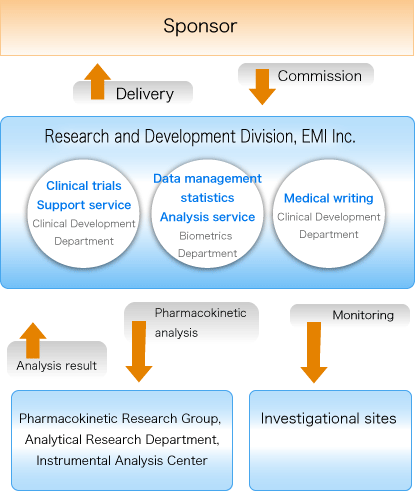
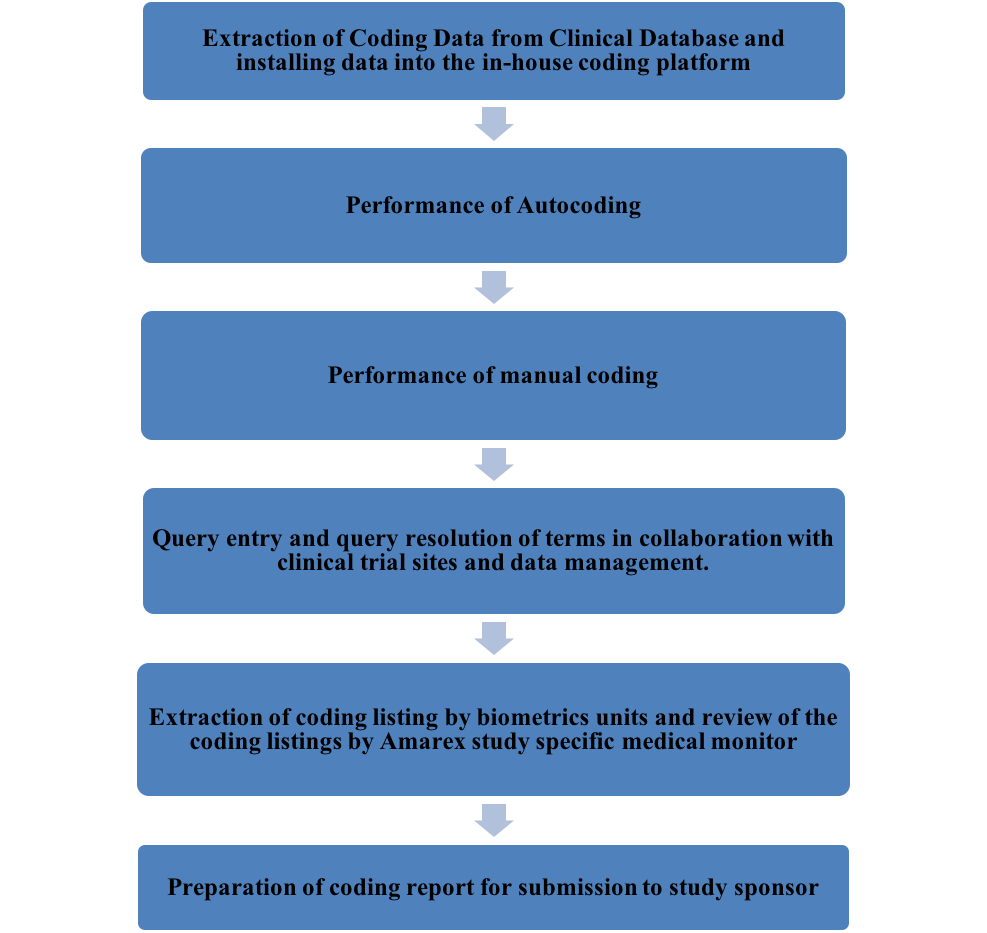
The importance of biometric functions to improving data quality in Clinical Trials | World Pharma Today
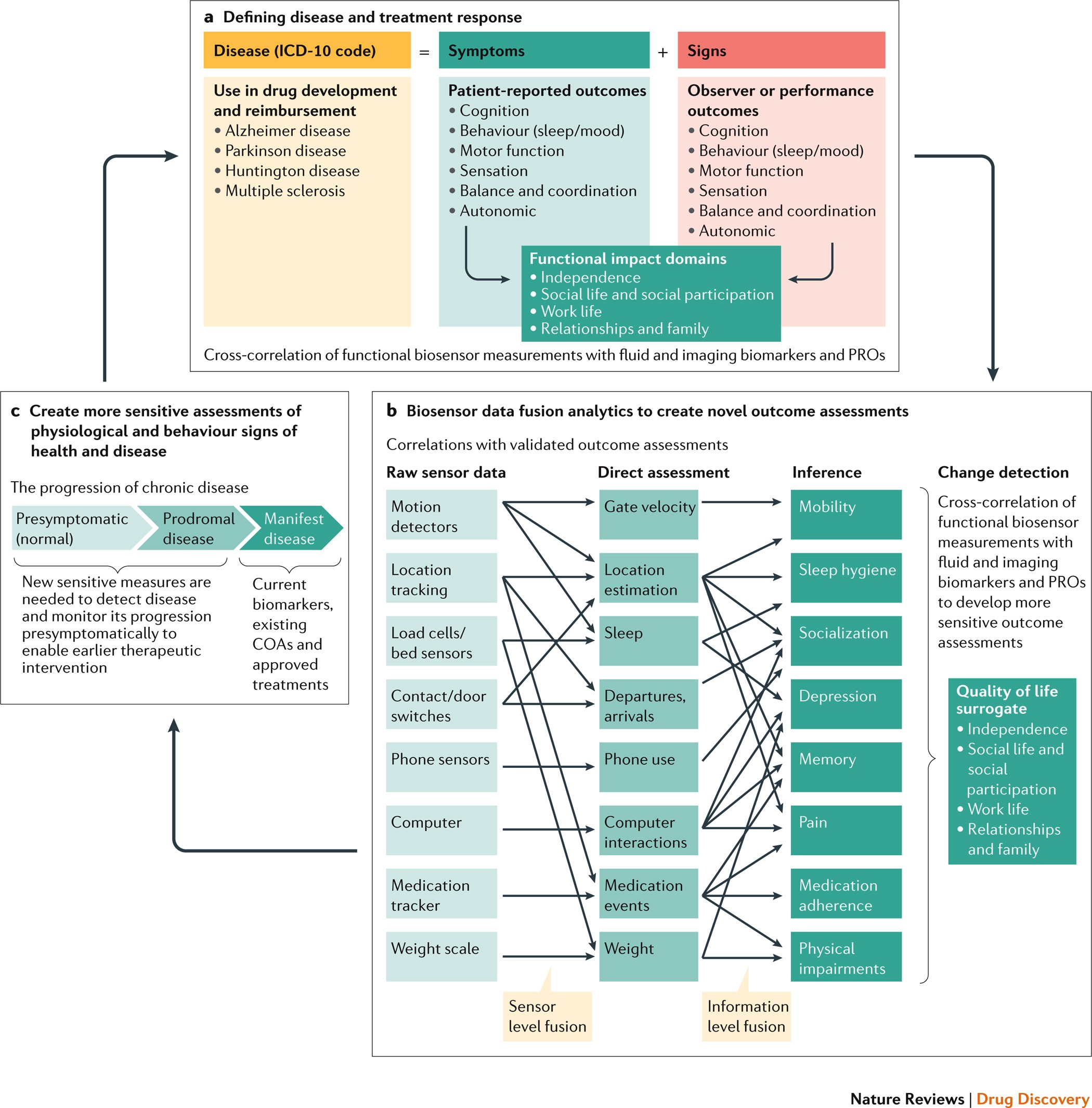
Biometric monitoring devices for assessing end points in clinical trials: developing an ecosystem | Nature Reviews Drug Discovery

Sample Size Calculations in Clinical Research 2nd edition by CHOW, S.‐C., SHAO, J., and WANG, H. - Glueck - 2008 - Biometrics - Wiley Online Library