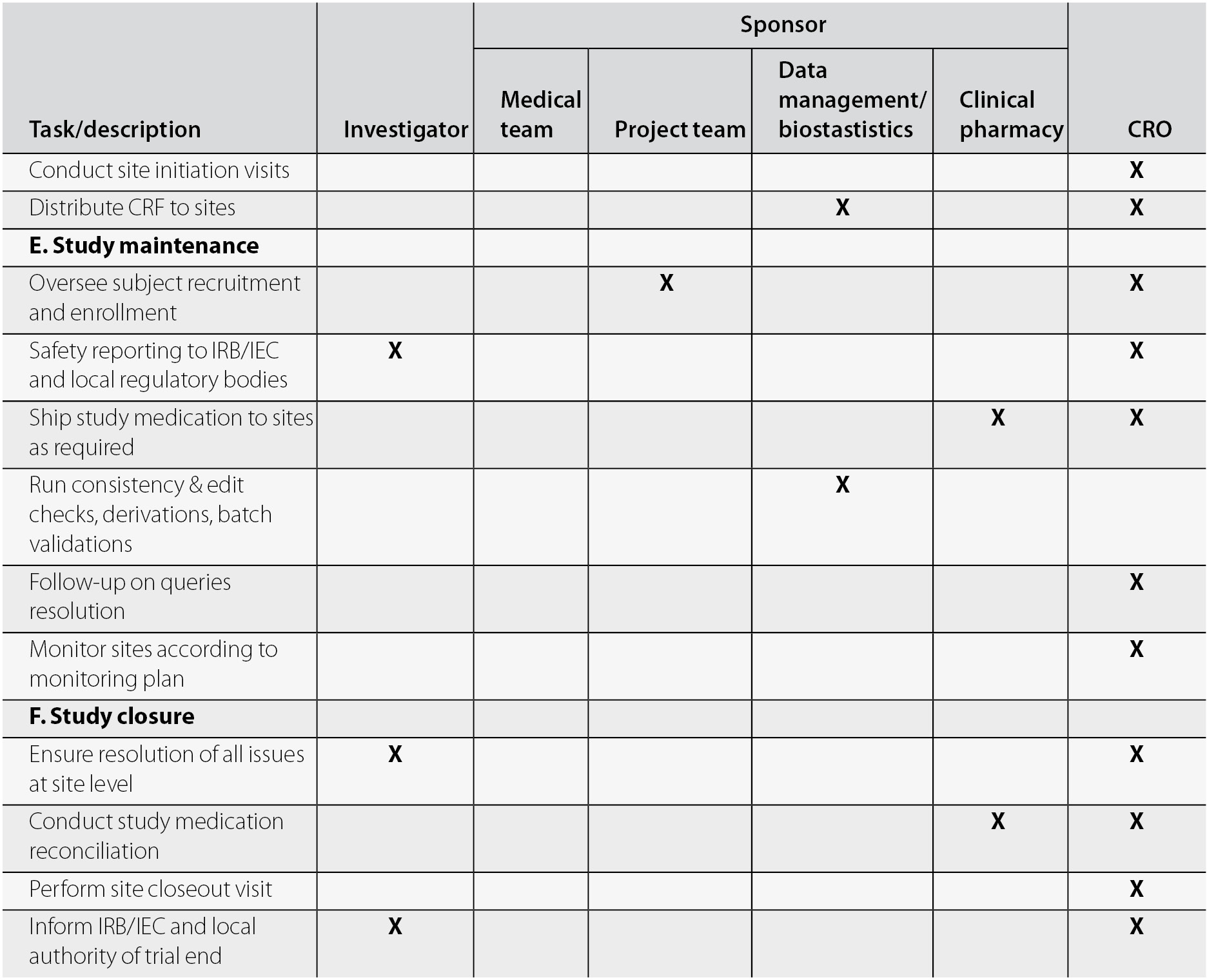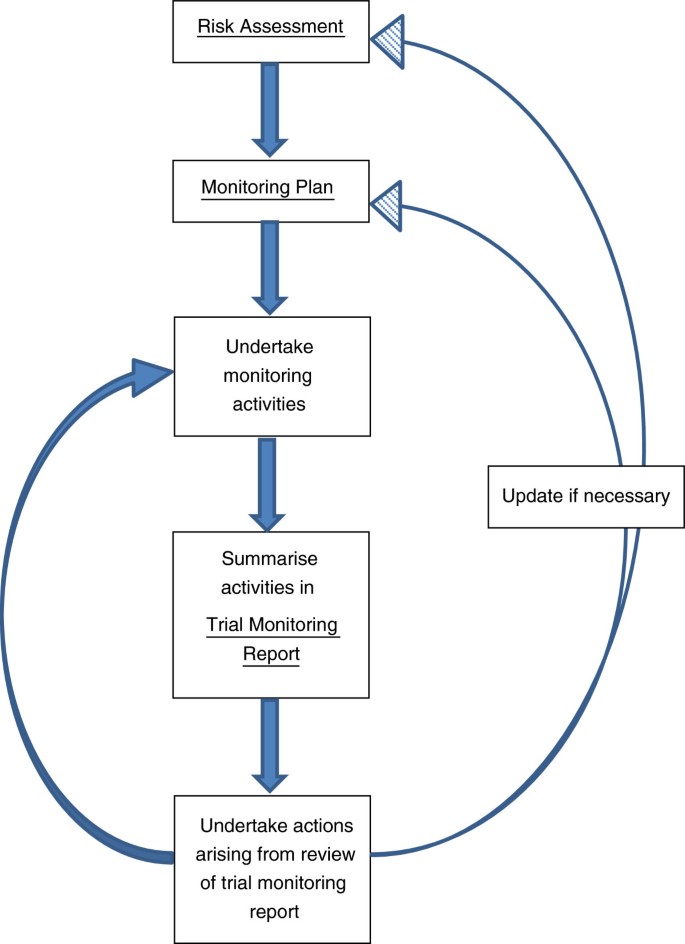
Risk-proportionate clinical trial monitoring: an example approach from a non-commercial trials unit | Trials | Full Text

Browse Our Example of Clinical Trial Safety Management Plan Template | Smartsheet, Report template, Best templates

Safety Management Plan – Clinical Trial Medical Monitoring Plan | Online Clinical Research Courses In India

Risk Management – Clinical Trial Medical Monitoring Plan | Online Clinical Research Courses In India
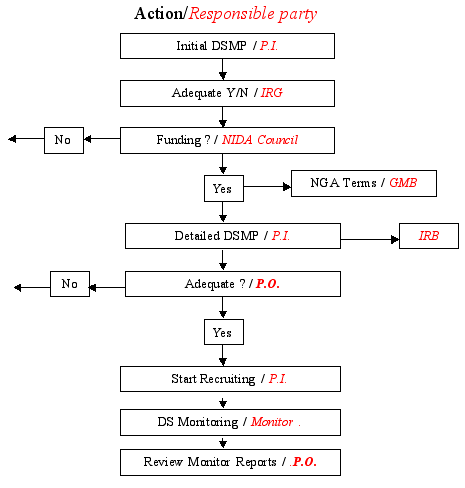
Guidelines for Developing a Data and Safety Monitoring Plan | National Institute on Drug Abuse (NIDA)

Guidelines for Developing a Data and Safety Monitoring Plan | National Institute on Drug Abuse (NIDA)

Phase I dose-escalation single centre clinical trial to evaluate the safety of infusion of memory T cells as adoptive therapy in COVID-19 (RELEASE) - eClinicalMedicine

Clinical Trial Safety Management Plan Ppt PowerPoint Presentation File Gridlines PDF - PowerPoint Templates

Risk Management – Clinical Trial Medical Monitoring Plan | Online Clinical Research Courses In India



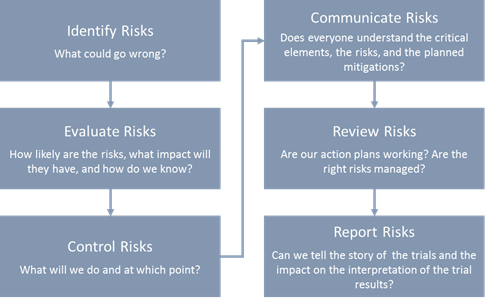



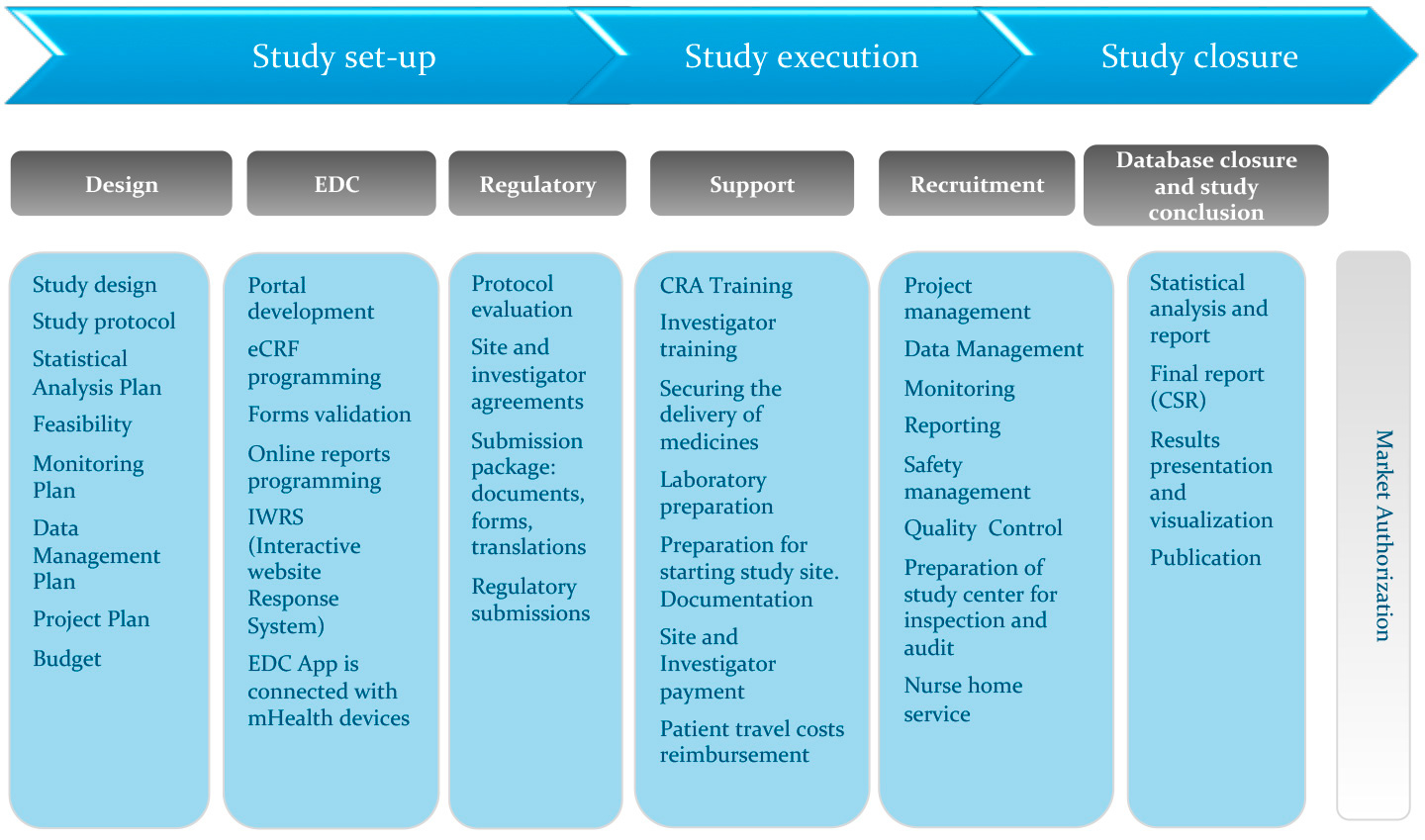



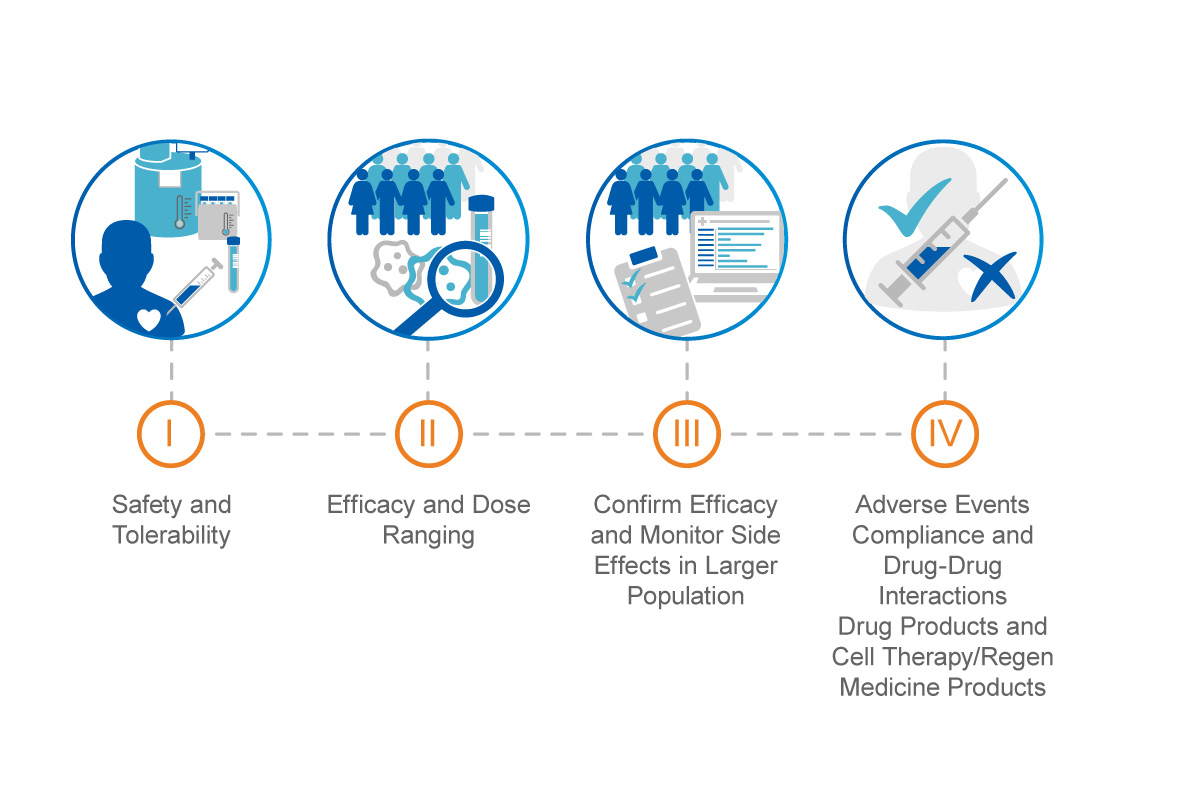
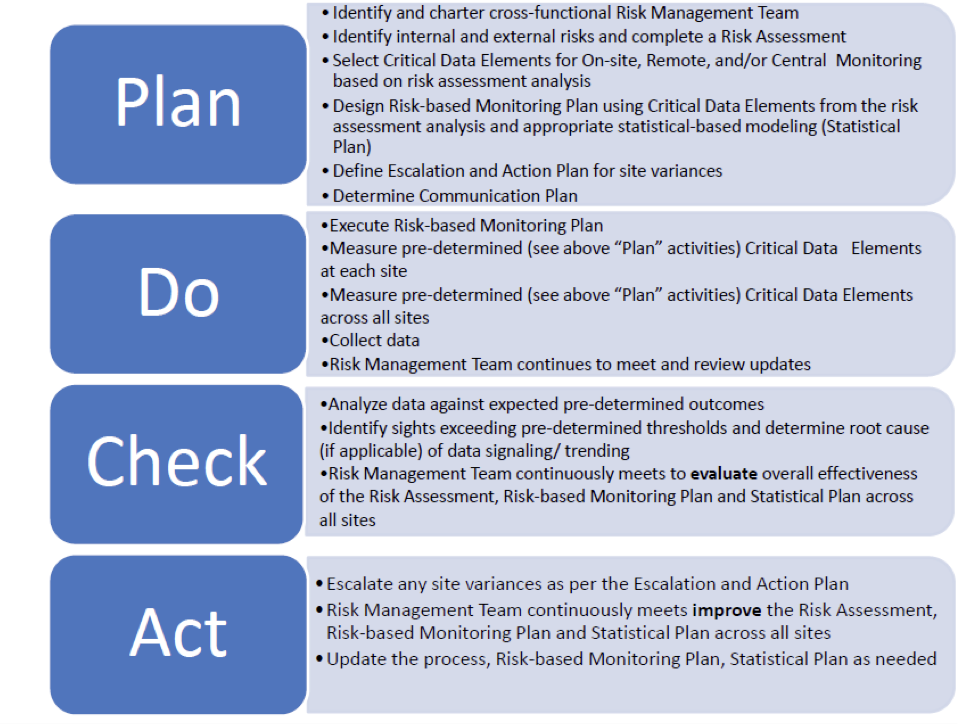
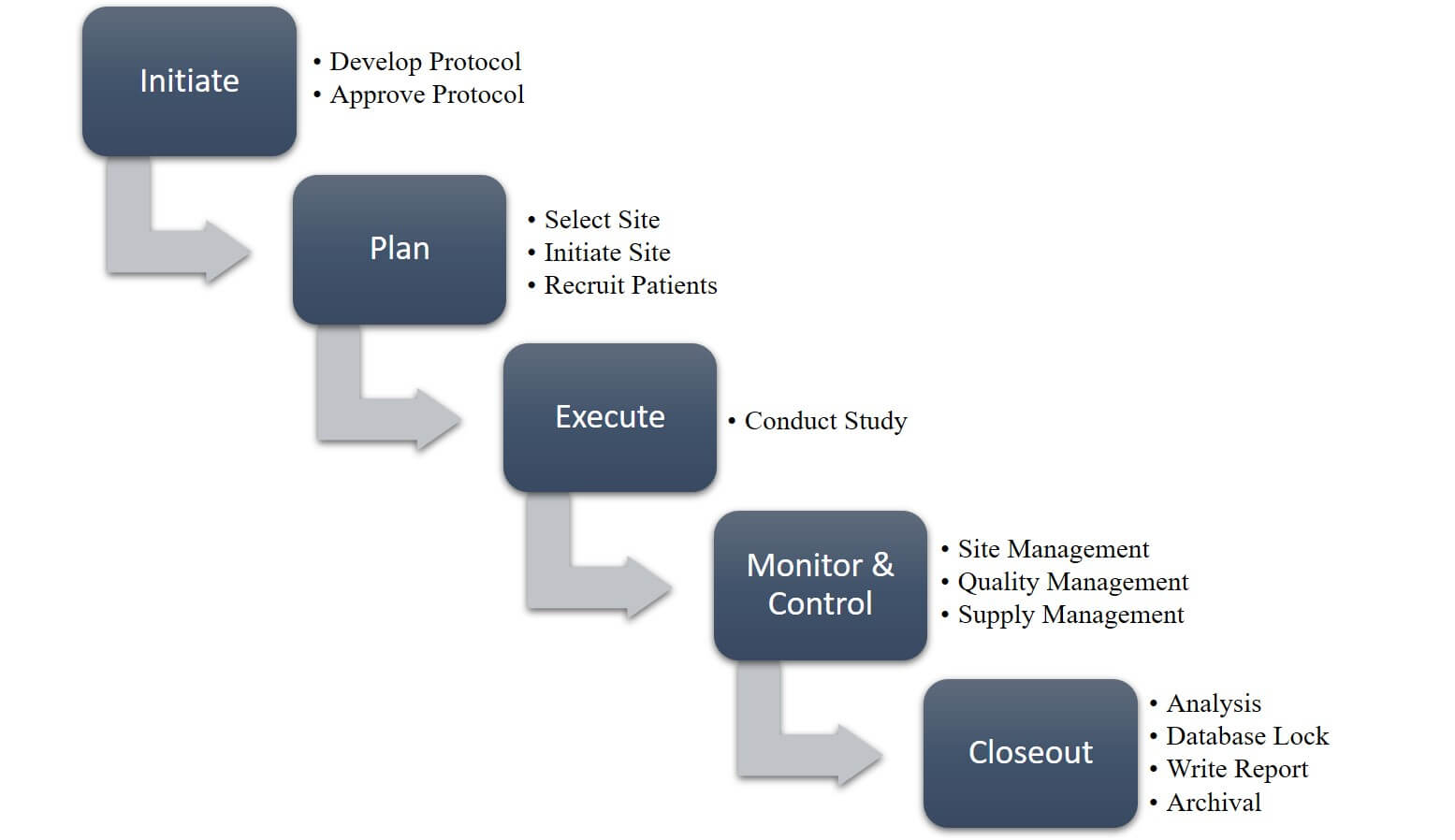
d9a3b07696026559b82aff0000ccbde2.jpg?sfvrsn=b9a1d5cd_0)