Gene therapy clinical trial protocols reviewed by the Recombinant DNA... | Download Scientific Diagram

PPT - Launching Clinical Trials: Small Company vs Large Company Environment PowerPoint Presentation - ID:6072994

Eligibility Criteria and Representativeness of Randomized Clinical Trials That Include Infants Born Extremely Premature: A Systematic Review - The Journal of Pediatrics

Clinical organizational chart for the LINCL study (BB IND 11481). The... | Download Scientific Diagram

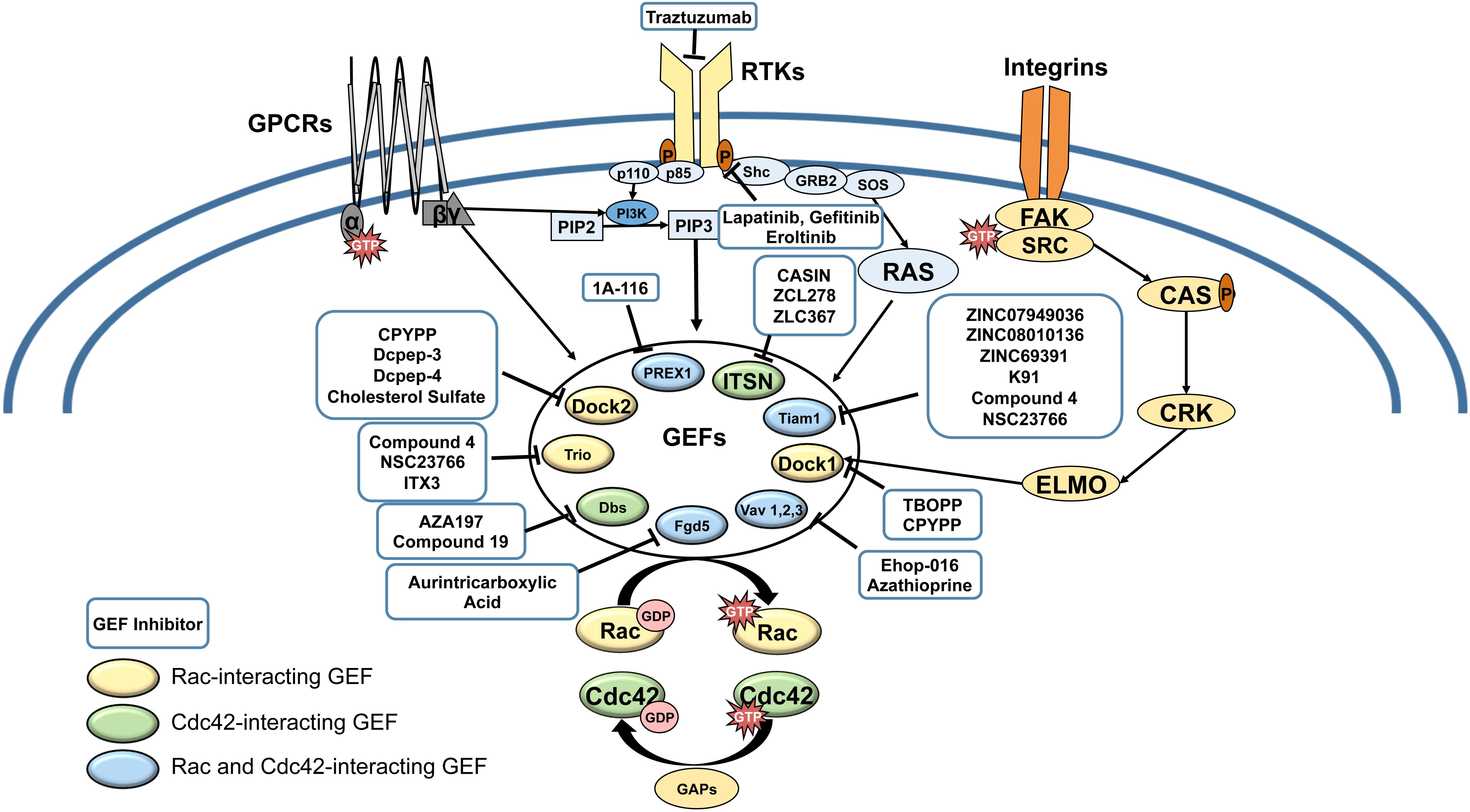
Comparison of Regulations for the Development of Oncolytic Virus Therapy in the United States, the European Union, and Japan. | Semantic Scholar

Gene and Virus Therapy Shared Resource - Mayo Clinic Comprehensive Cancer Center Research - Mayo Clinic Research

Race Oncology confirms positive results from Bisantrene drug trial on patients with advanced acute myeloid leukaemia

Efficacy and safety of hydroxychloroquine as pre-and post-exposure prophylaxis and treatment of COVID-19: A systematic review and meta-analysis of blinded, placebo-controlled, randomized clinical trials. - The Lancet Regional Health – Americas