Clinical Trials: Study Design, Endpoints and Biomarkers, Drug Safety, and FDA and ICH Guidelines by Tom Brody PhD | NOOK Book (eBook) | Barnes & Noble®

Clinical Trials: Study Design, Endpoints and Biomarkers, Drug Safety, and FDA and ICH Guidelines - Kindle edition by Brody, Tom. Professional & Technical Kindle eBooks @ Amazon.com.

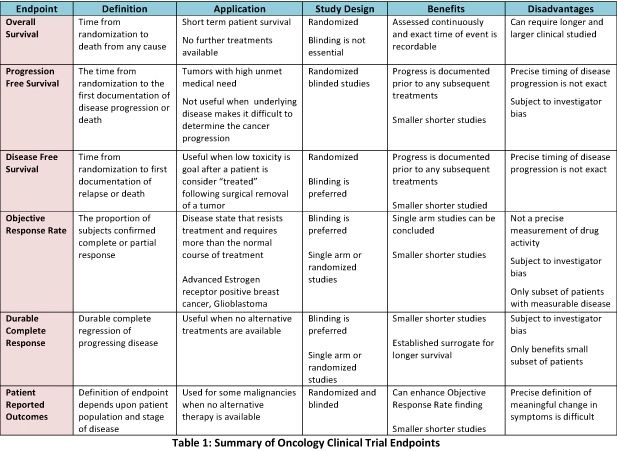
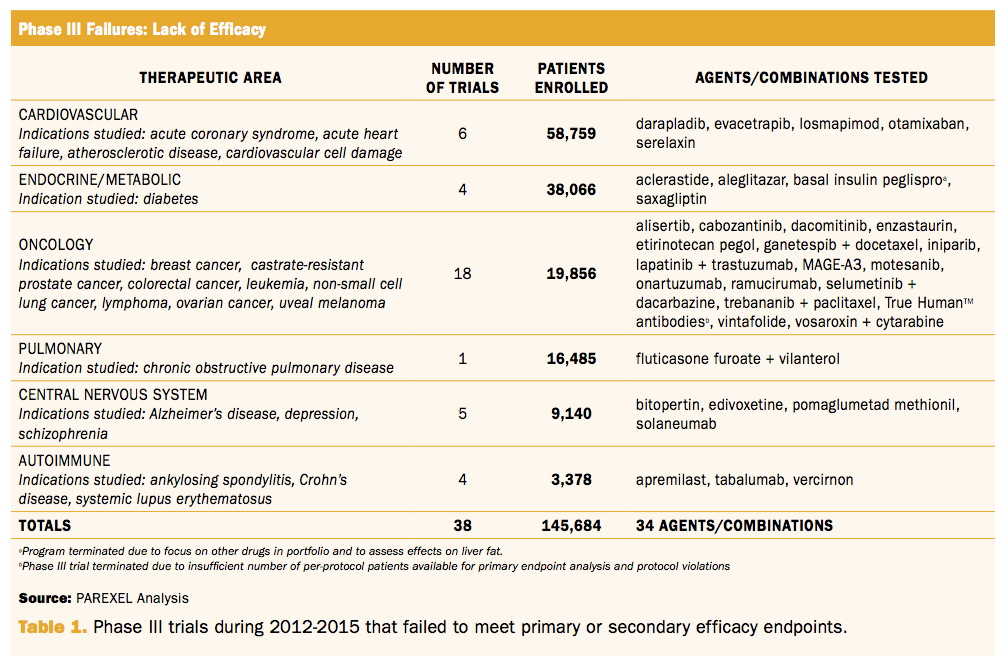
Challenging Issues in Clinical Trial Design: Part 4 of a 4-Part Series on Statistics for Clinical Trials - ScienceDirect

Astegolimab (anti-ST2) efficacy and safety in adults with severe asthma: A randomized clinical trial - Journal of Allergy and Clinical Immunology
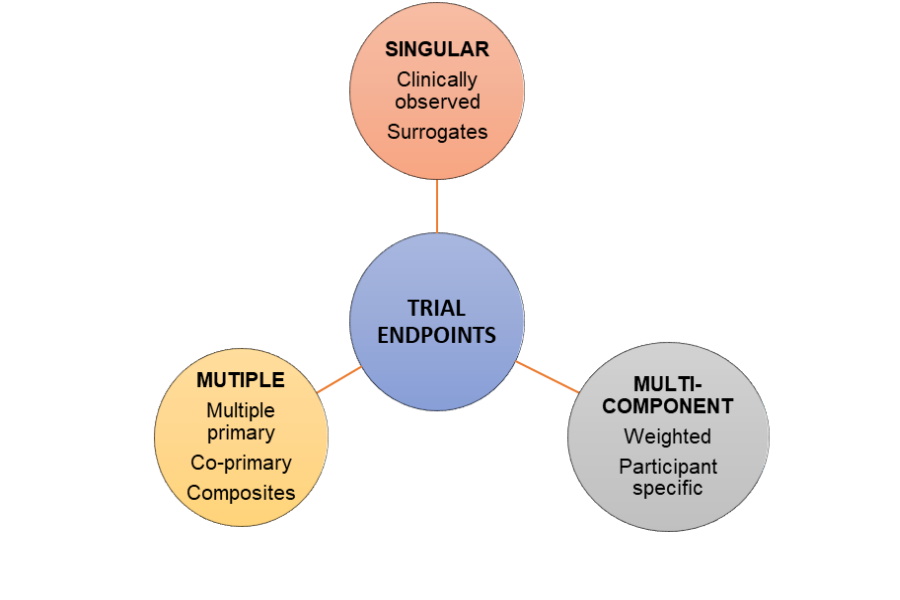
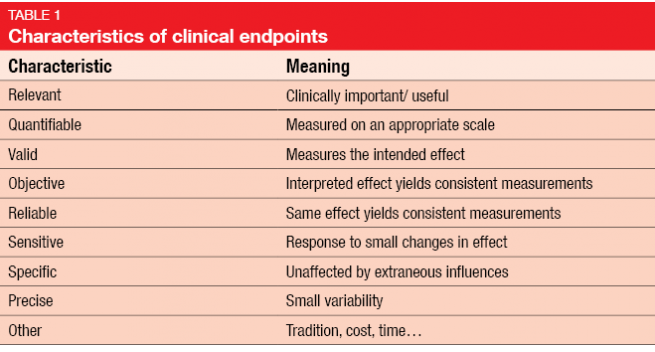
The Endpoint Selection: a Complex Process in the Clinical Trials Design Page CRA School | The International Clinical Research Academy Page | CRA School | The International Clinical Research Academy
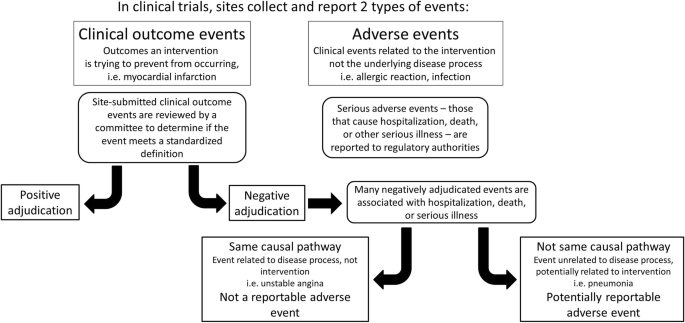
Methods for safety and endpoint ascertainment: identification of adverse events through scrutiny of negatively adjudicated events | Trials | Full Text
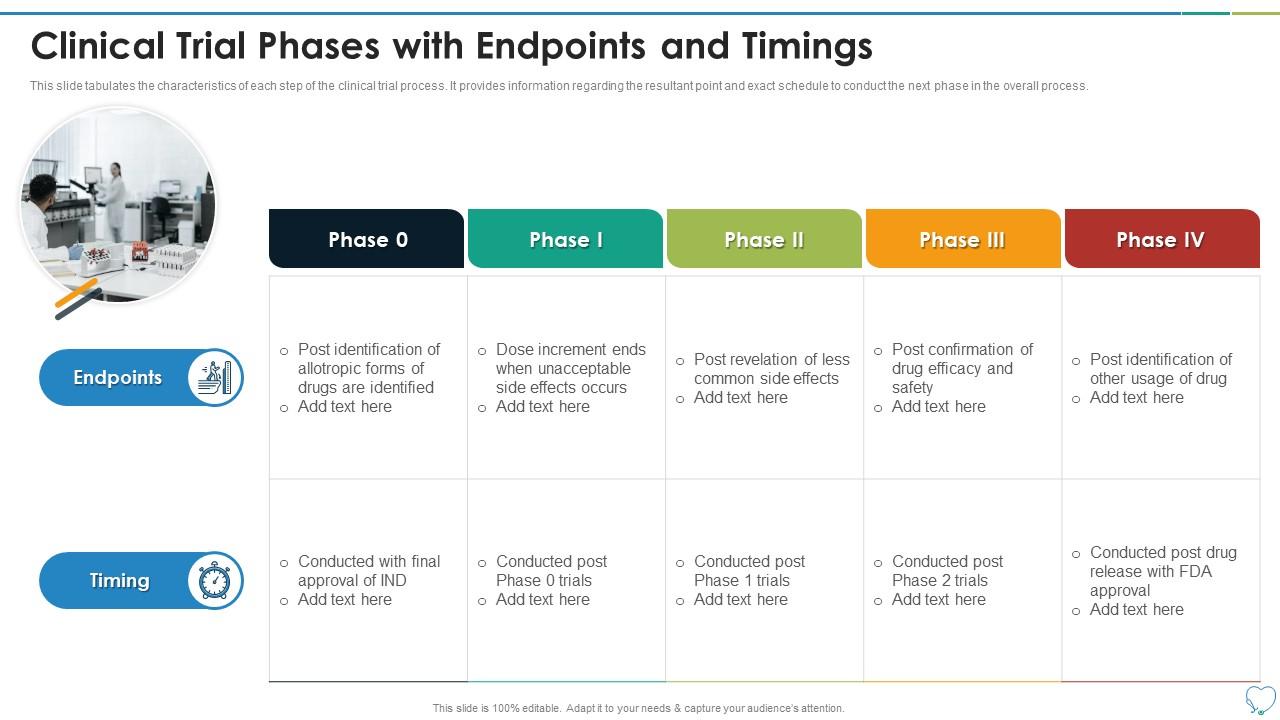
Clinical Trial Phases Endpoints And Timings | Presentation Graphics | Presentation PowerPoint Example | Slide Templates
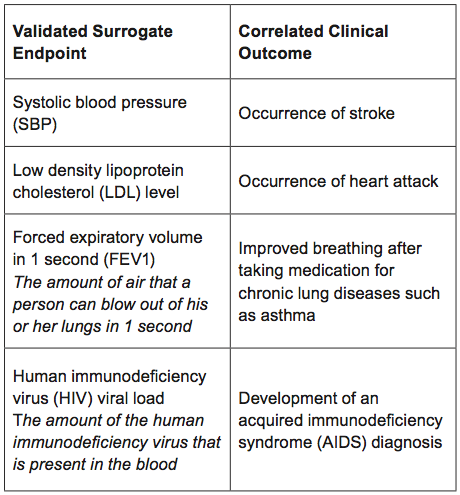
The use of validated and nonvalidated surrogate endpoints in two European Medicines Agency expedited approval pathways: A cross-sectional study of products authorised 2011–2018 | PLOS Medicine





![Stages of PWS Drug Development Overview [VIDEO] Stages of PWS Drug Development Overview [VIDEO]](https://www.fpwr.org/hubfs/Clinical%20Trials/Drug%20Development%20Process/Slide5.jpeg)